2022年3月15日
COVID-19論文 アップデート 2021.02.04 報
【ワクチン】
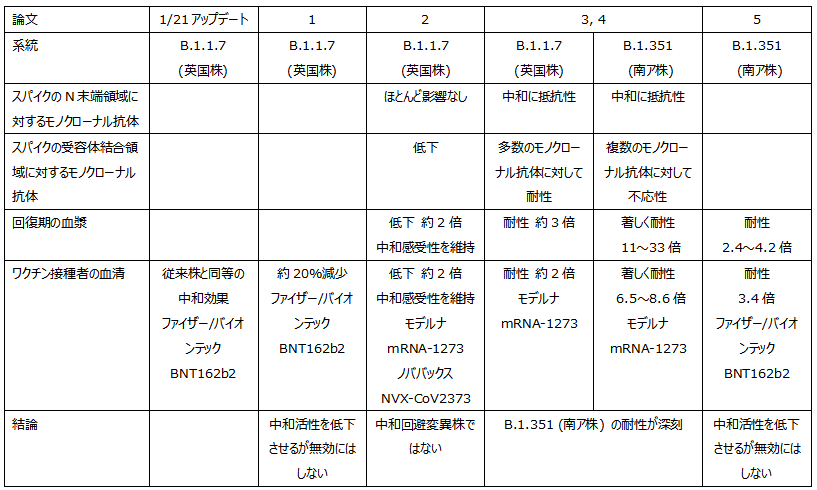
1/21アップデート
Shi PY, et al. Neutralization of N501Y mutant SARS-CoV-2 by BNT162b2 vaccine-elicited sera. Preprint. Res Sq. 2021;rs.3.rs-143532. doi:10.21203/rs.3.rs-143532/v1
Xie X, et al. Neutralization of N501Y mutant SARS-CoV-2 by BNT162b2 vaccine-elicited sera. Preprint. bioRxiv. 2021;2021.01.07.425740. doi:10.1101/2021.01.07.425740ファイザー/BioNTech テキサス大学のグループによる有効性の確認
ファイザー/バイオンテックのmRNAワクチンBNT162b2接種者の血清は、N501Y変異株に対して従来株と同等の中和効果を示す
- Muik A, et al. Neutralization of SARS-CoV-2 lineage B.1.1.7 pseudovirus by BNT162b2 vaccine-elicited human sera. Science. 2021;eabg6105. doi:10.1126/science.abg6105
- ファイザー/バイオンテックのグループによる有効性のチェック
- mRNAワクチンBNT162b2接種者の血清 B.1.1.7系統 (英国株) に対する中和活性は約20%減少
- インフルエンザワクチンからの推測では、20%程度の減少は生物学的に有意ではない
- Shen X, Tang H, McDanal C, et al. SARS-CoV-2 variant B.1.1.7 is susceptible to neutralizing antibodies elicited by ancestral Spike vaccines. Preprint. bioRxiv. 2021;2021.01.27.428516. doi:10.1101/2021.01.27.428516
- Ho D, et al. Increased Resistance of SARS-CoV-2 Variants B.1.351 and B.1.1.7 to Antibody Neutralization. Preprint. Res Sq. 2021;rs.3.rs-155394. doi:10.21203/rs.3.rs-155394/v1
- Wang P, et al. Increased Resistance of SARS-CoV-2 Variants B.1.351 and B.1.1.7 to Antibody Neutralization. Preprint. bioRxiv. 2021;2021.01.25.428137. doi:10.1101/2021.01.25.428137
- Jangra S, et al. The E484K mutation in the SARS-CoV-2 spike protein reduces but does not abolish neutralizing activity of human convalescent and post-vaccination sera. Preprint. medRxiv. 2021;2021.01.26.21250543. doi:10.1101/2021.01.26.21250543
- Logunov DY, et al. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomised controlled phase 3 trial in Russia. Lancet. DOI:https://doi.org/10.1016/S0140-6736(21)00234-8
- ロシアのアデノウイルスベクターDNAワクチンGam-COVID-Vac(スプートニクV)のP3試験の中間結果
- ワクチン群14 964人→16人 (0・1%) 感染 プラセボ群4902人→62人 (1.3%) 感染 ワクチンの有効性: 91・6%
- 報告された有害事象の94・0%は軽微
- 重篤な有害事象: ワクチン群の0・3%, プラセボ群の0・4%, ワクチン接種に関連すると見なされたものはない
- 死亡: ワクチン群3人, プラセボ群1人, ワクチン接種に関連すると見なされていない
- P3試験途中でロシア政府が承認に踏みきったため安全性や品質に懸念が出ていたが、特に問題はなさそうである
- Mahase E. Covid-19: Novavax vaccine efficacy is 86% against UK variant and 60% against South African variant. BMJ. 2021;372:n296. doi:10.1136/bmj.n296
- Novavax 社のタンパク質ナノ粒子ワクチンNVX-CoV2373のP3試験の中間結果 18~84才の参加者15000人以上 有効性: 従来株 95.6%, B.1.1.7系統 (英国株) 85.6%, B.1.351系統 (南ア株) 60%
- Janssen社/Johnson & Johnson社のDNAワクチンAd26.COV2.SのP3試験の中間結果 参加者43783人 ワクチン接種後28日以降の中等症~重症COVID-19の予防率: 米国72%、中央・南アメリカ66%、南アフリカ57%、全体66% B.1.351系統からのSARS-CoVの-2変異体を含む地域を含んでいる 単回投与でよい
【治療薬】
- Dai Y, et al. A large-scale transcriptional study reveals inhibition of COVID-19 related cytokine storm by traditional chinese medicines. Sci Bull (Beijing). 2021;10.1016/j.scib.2021.01.005. doi:10.1016/j.scib.2021.01.005 中国科学院と中国国家自然科学財団が共同出版するジャーナル
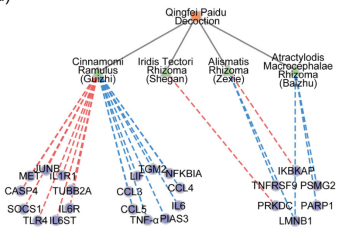
- 578種類の漢方生薬 国家衛生健康委員会や各省・市からリリースされている治療プロトコル53種類から収集した抗COVID-19漢方処方338種類(伝統的処方196, 新規開発処方142) → 培養免疫細胞 → 遺伝子発現の変化を網羅的解析 → IL-6シグナルを標的とする16種類, TNF-αシグナルを標的とする37種類を選択→ さらに詳細なネットワーク薬理学 下図: 清肺排毒湯の処方ハーブー遺伝子ネットワーク(赤線: 遺伝子発現↑, 青線: 遺伝子発現↓)
- Li Y, et al. Potential effect of Maxing Shigan decoction against coronavirus disease 2019 (COVID-19) revealed by network pharmacology and experimental verification. J Ethnopharmacol. 2021;271:113854. doi:10.1016/j.jep.2021.113854
- 麻杏石甘湯(麻杏甘石湯)のネットワーク薬理学
- 有効成分105, ターゲットタンパク質1025
- COVID-19に関連する83のターゲットタンパク質, 63のシグナル伝達経路
- IL-6シグナル伝達経路に関連するタンパク質の発現レベルを効果的に阻害
- ACE2、Mpro、RdRpとの親和性が強い成分は、主に甘草と苦杏仁に由来 (アミグダリン、ユークレノン、グリチルリチン、グリシロール)
- Xiong H, et al. Analysis of the mechanism of Shufeng Jiedu capsule prevention and treatment for COVID-19 by network pharmacology tools. Eur J Integr Med. 2020;40:101241. doi:10.1016/j.eujim.2020.101241
- 疎風解毒カプセルのネットワーク薬理学
- 化合物163種類, 標的遺伝子463種類
- 複数の化合物に共通して標的とされるタンパク質76種類
- 作用の中心となる標的タンパク質: TNF、IL-10、IL-2、IL-6、STAT1、CCL2 ← ケルセチン、ルテオリン、ウォゴニン、ケンフェロール
- Zhu YW, et al. Analyzing the potential therapeutic mechanism of Huashi Baidu Decoction on severe COVID-19 through integrating network pharmacological methods. J Tradit Complement Med. 2021;10.1016/j.jtcme.2021.01.004. doi:10.1016/j.jtcme.2021.01.004
- 化湿敗毒湯のネットワーク薬理学
- 含有化合物1615種類, 有効成分204種類,
- 標的タンパク質259種類
- COVID-19に関連する可能性がある標的遺伝子45種類 ハブとなる標的遺伝子13種類: RELA、TNF、IL6、IL1B、MAPK14、TP53、CXCL8、MAPK3、MAPK1、IL4、MAPK8、CASP8 、STAT1
- 標的となるシグナル伝達経路: 炎症および免疫調節経路
- 抗COVID-19作用の中心となる生薬: 甘草・麻黄・蒼朮・黄耆 抗COVID-19作用の中心となる有効成分: ケルセチン、ケンフェロール、β-シトステロール、イソラムネチン、ナリンゲニン、ルテオリン、カテキン、デルフィニジン、アロエエモジン、バイカレイン、イリソリドン
- Ceccarelli G, et al. Oral Bacteriotherapy in Patients With COVID-19: A Retrospective Cohort Study. Front Nutr. 2021;7:613928. doi:10.3389/fnut.2020.613928
- 重症COVID-19患者
- 標準治療(低分子量ヘパリン+ヒドロキシクロロキン・抗ウイルス薬・トシリズマブうち1つ以上)のみ: 112人 標準治療+経口細菌療法88人
- 死亡率: 標準治療のみ群30% vs. 標準治療+経口細菌療法群11%
- Sivomixx®: 8種類の乳酸菌をブレンドした市販サプリ (Streptococcus thermophilus DSM 32245, Bifidobacterium lactis DSM 32246, Bifidobacterium lactis DSM 32247, Lactobacillus acidophilus DSM 32241, Lactobacillus helveticus DSM 32242, Lactobacillus paracasei DSM 32243, Lactobacillus plantarum DSM 32244, Lactobacillus brevis DSM 27961)
- Schultz IC, et al. Mesenchymal Stem Cell-Derived Extracellular Vesicles Carrying miRNA as a Potential Multi Target Therapy to COVID-19: an In Silico Analysis. Stem Cell Rev Rep. 2021;1-16. doi:10.1007/s12015-021-10122-0
- 間葉系幹細胞由来の細胞外小胞に含まれているマイクロRNAが、サイトカイン・ケモカイン制御障害、細胞死 (=組織傷害) および凝固障害を調節する能力を持つかどうかを解析
- 骨髄、臍帯、脂肪組織由来の各間葉系幹細胞の細胞外小胞に含まれるマイクロRNAをNGS解析
- 58種類のマイクロRNAが重複
- ターゲットmRNAを解析 → サイトカイン・ケモカイン制御に関連する遺伝子をターゲットとするマイクロRNA: 258 細胞死に関連する遺伝子をターゲットとするマイクロRNA: 266 凝固カスケードに関連する遺伝子をターゲットとするマイクロRNA: 148
- 間葉系幹細胞由来の細胞外小胞に含まれているマイクロRNAは、重度のCOVID-19患者の生存率を改善できる可能性がある

【検査】
- Okoye NC, et al. Performance Characteristics of BinaxNOW COVID-19 Antigen Card for Screening Asymptomatic Individuals in a University Setting. J Clin Microbiol. 2021;JCM.03282-20. doi:10.1128/JCM.03282-20
- アボットBinaxNOWCOVID-19抗原検査キットの評価
- 無症候ユタ大学生2645人
- RT-PCR陽性者: 1.7% (45/2645)
- BinaxNOW: 24/RT-PCR陽性者45, 感度53.3%, 特異度100%
- RT-PCR陽性かつBinaxNOW陽性のCt値は、RT-PCR陽性かつBinaxNOW陰性と比較して有意に低い (17.6 vs 29.6)
- ウイルス量が多い(Ct <23.0)個人では、RT-PCRとBinaxNOWの一致率95.8%
- 偽陰性の可能性が高いため、無症候性の個人をスクリーニングするために迅速抗原検査を利用する場合は注意が必要
【その他】
- Landoni G, et al. Nations with high smoking rate have low SARS-CoV-2 infection and low COVID-19 mortality rate. Acta Biomed. 2020;91(4):e2020168. doi:10.23750/abm.v91i4.10721
- COVID-19の死亡率 男性喫煙者>45%の国: 13/100万人 vs. 男性喫煙者<25%未満の国: 33/100万人
- SARS-CoV-2感染率 男性喫煙者>45%の国: 436/100万人 vs. 男性喫煙者<25%未満の国: 1139/100万人
- 能動喫煙はCOVID-19に対して保護的
- Marouf N, et al. Association between periodontitis and severity of COVID-19 infection: a case-control study. J Clin Periodontol. 2021;10.1111/jcpe.13435. doi:10.1111/jcpe.13435
- COVID-19患者568人
- 歯周炎のある患者は、死亡リスク8.81倍、ICU入院リスク3.54倍、要人工呼吸器リスク4.57倍
- 歯周炎のある患者は、COVID-19の重症化に関連する白血球、D-ダイマー、CRPの血中濃度が有意に高い

- Asher A, et al. Blood omega-3 fatty acids and death from COVID-19: A pilot study [published online ahead of print, 2021 Jan 20]. Prostaglandins Leukot Essent Fatty Acids. 2021;166:102250. doi:10.1016/j.plefa.2021.102250
- ω-3インデックス(血中EPA+DHA)が死亡リスクと逆相関するという仮説をテストするためのパイロット研究
- 患者100人のうち死亡14人 ω-3インデックス中位75人のうち死亡13人 ω-3インデックス上位25人 (ω-3インデックス≥5.7%) のうち死亡1人 → 中位に対する死亡リスク0.25 (p=0.07)
- Kalarikkal SP, et al. Edible plant-derived exosomal microRNAs: Exploiting a cross-kingdom regulatory mechanism for targeting SARS-CoV-2. Toxicol Appl Pharmacol. 2021;115425. doi:10.1016/j.taap.2021.115425
- 食用植物由来のエクソソーム様小胞は、食用ナノ粒子(edible nanoparticles: ENPs)として知られている ENPsは多種多様なマイクロRNAを含んでいる 植物由来のマイクロRNAがヒトmRNAを制御する「界を越えた調節」が実証されている ENPsを投与すると肺組織に蓄積することが知られている → SARS-CoV-2のmRNAに対するマイクロRNAを含むENPsがあれば、COVID-19治療に使える可能性がある
- データベースから取得したENPs由来マイクロRNA260種類 → SARS-CoV-2のmRNAを標的とする可能性のある22種類を特定 (大豆、ショウガ、ハミウリ、グレープフルーツ、トマト、ナシ)
- SARS-CoV-2を標的とするENP由来マイクロRNAを治療に利用できる可能性がある
【参考】 ENPs
- 食用植物由来のエクソソーム様小胞
- サイズ: 100~900nm (哺乳類の細胞外小胞のサイズ: エクソソーム40~120nm, 微小小胞体50~1000nm, アポトーシス小体500~2000nm)
- 構造・機能的に哺乳類のエクソソームに類似
- 植物内での由来は不明
- 小胞内に植物由来の核酸(DNA, mRNA, マイクロRNAなど)・タンパク質を含む
- ヒトのmRNAを標的とすることが予測されている多数のマイクロRNAも含まれている → 界を越えてヒトにも作用し得る
- 様々な食用植物 (ブルーベリー、ココナッツ、ショウガ、グレープフルーツ、ハミウリ、キウイフルーツ、オレンジ、エンドウ豆、ナシ、大豆、トマト、ブドウ) のENPsからそれぞれ30〜130種類程度のマイクロRNAが同定されている
- ショウガENPsをマウス鼻腔および静脈に投与 → 肺組織に移行し、投与後72時間まで検出可能
- ショウガ由来ENPsに存在するマイクロRNA → 腸内細菌叢L. rhamnosusのモノオキシゲナーゼmRNAを抑制 → 宿主の腸管免疫を調節
- クルミ由来ENPsに存在するマイクロRNA → 脂肪細胞のTNF-αシグナル伝達経路を調節 → 炎症を調節

